microRNA-218 promotes gemcitabine sensitivity in human pancreatic cancer cells by regulating HMGB1 expression
Introduction
Currently, malignant tumors are a part of major diseases that are harmful to human health. Pancreatic cancer is a type of gastrointestinal tract malignancy that typically has a poor prognosis (1). In China, pancreatic cancer is one of the three leading causes of gastrointestinal tract cancer-related deaths, and the mortality rate of pancreatic cancer continues to show an upward trend year by year (2-4). Surgical resection is the only hope for improving the survival rate of patients with pancreatic cancer. However, 85% of these patients suffer from advanced-stage pancreatic cancer at the time of seeking treatment. Surgical treatment is possible in only 10-15% of pancreatic cancer patients, and the average 5-year survival rate after surgery for these patients is less than 5% (1,5). In addition to surgery, chemotherapy remains one of the important means of treating advanced-stage pancreatic cancer, preventing postoperative recurrence, prolonging patient survival time, and improving the quality of life for the patients (6,7).
2',2'-Difluorodeoxycytidine (dFdC; also known as gemcitabine, GEM) is a structural analogue of a DNA nucleotide. Under the action of nucleotide kinase, GEM is converted to active gemcitabine diphosphate (dFdCDP) and gemcitabine triphosphate (dFdCTP) (8,9). The metabolite of GEM not only blocks cellular DNA synthesis through competing with deoxycytidine but also inhibits the production of deoxycytidine (10). GEM pairs with multiple nucleotides (NT) and exhibits a different terminal structure. DNA exonuclease is incapable of recognizing the terminal structure of GEM. As the result, DNA exonuclease-mediated DNA repair is inhibited, which induces apoptosis in the S phase of the cell cycle (DNA synthesis phase) via the caspase cascade amplification system (11,12). Phase m clinical trials have shown that compared with 5-fluorouracil, GEM significantly improves the symptoms in patients and prolongs the median survival time of patients. Therefore, GEM has become the preferred chemotherapy drug for advanced pancreatic cancer (13-16). However, it has been found during long-term clinical practice that the efficacy of GEM in treatment of pancreatic cancer is far from ideal, with the overall efficacy of GEM being less than 20%. The main reason lies in the acquired or intrinsic drug resistance of pancreatic cancer cells, which leaves many GEM-treated patients to suffer the side effects of chemotherapy without obtaining satisfactory therapeutic effects (17-19). Therefore, we are facing great clinical and experimental challenges, which include reducing the resistance of pancreatic cancer to GEM, exploring the mechanism responsible for reversal of GEM resistance in pancreatic cancer, and enhancing the efficacy of GEM (20).
MicroRNAs (miRNAs) are a class of endogenous, non-coding, single-stranded small RNA molecules approximately 22 NT in length. miRNAs regulate mRNA expression at the posttranscriptional level through complementary pairing with the 3’ untranslated region (3' UTR) of the target gene mRNA, thus participating in physiological processes, such as cell proliferation, apoptosis, differentiation, metabolism and development, as well as pathological processes, such as cardiovascular diseases, neurological diseases and cancer (21-23). It is predicted that more than one-third of the human genes are target genes for conserved miRNAs. The role of miRNAs in human diseases, especially tumors, has received increasing attention (24,25).
Studies on the relationship between miRNAs and tumor chemosensitivity have attracted increasing attention from scholars in the field of cancer research and have become a research hotspot in recent years. Previous studies have reported that microRNA-218 (miR-218) is a tumor-suppressing microRNA. A study conducted by Li et al. has shown that miR-218 inhibits the growth of cervical cancer and enhances the sensitivity of cervical cancer to cisplatin-based chemotherapy (26). Our previous study has shown that miR-218 expression is downregulated in human pancreatic cancer tissues and cell lines. miR-218 negatively regulates the expression of high mobility group box 1 (HMGB1) protein and inhibits the proliferation and invasion of pancreatic cancer cells. However, the relationship between miR-218 and the sensitivity of pancreatic cancer cells to GEM-based chemotherapy has not been reported. This study mainly investigated the relationship between miR-218 and the sensitivity of pancreatic cancer cells to GEM and explored the signaling pathways involved, thus providing novel approaches and strategies for treatment of pancreatic cancer.
Materials and methods
Main reagents
The TaqMan miRNA Isolation Kit, TaqMan microRNA Assay Kit, TaqMan microRNA Assay and TaqMan Universal PCR Master Mix were purchased from Applied Biosystems—Life technologies, Inc (Waltham, MA, USA). The miR-218 RT primers, miR-218 mimic and non-specific control (mimic ctrl) were synthesized by Shanghai GenePharma Co., Ltd. HMGB1 siRNA and siRNA control (scramble) (siRNA ctrl) were synthesized by Shanghai GenePharma Co., Ltd. The primers required for construction of the recombinant HMGB1 expression vector were synthesized and provided by Shanghai Invitrogen Biotechnology Co., Ltd., which included the following primers: the upstream primer, 5'-CG GAA TTC ATG GGC AAA GGA GAT CCT AA-3' (containing the EcoR I restriction site); and the downstream primer, 5'-CG GGA TCC TTC ATC ATC ATC ATC TTC TT-3' (containing the BamH I restriction site). The EcoR I and BamH I restriction endonucleases were purchased from TaKaRa Biotechnology (Dalian) Co., Ltd. Taq DNA polymerase was purchased from Fermentas, Inc. (Glen Burnie, MD, USA).
The GEM-sensitive pancreatic cancer cell line, BxPC-3, and the GEM-resistant pancreatic cancer cell line, PANC-1, were purchased from American Type Culture Collection (ATCC, MD, USA). Fetal bovine serum (FBS), Dulbecco's Modified Eagle’s medium (DMEM), Roswell Park Memorial Institute (RPMI) 1640 medium, L-glutamine, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), Lipofectamine 2000, and Trizol reagent were purchased from Invitrogen (USA). Cell culture plates or dishes were purchased from Corning Inc. The Reverse Transcription Kit was purchased from Qiagen. The pGEM-T and pcDNA3.1 vectors were purchased from Invitrogen (USA).
The Cell Counting Kit-8 (CCK-8) was purchased from Beyotime Institute of Biotechnology (Shanghai, China). Trypsin and phosphate buffered saline (PBS) were purchased from Sigma-Aldrich (USA). Annexin V and propidium iodide (PI) were purchased from Hoffmann-La Roche Ltd. The primary antibodies, including the rabbit anti-human HMGB1 monoclonal antibody and the mouse anti-human β-actin monoclonal antibody, were purchased from Abcam (UK). The secondary antibodies, including horseradish peroxidase (HRP)-conjugated affinity-purified goat anti-mouse IgG and HRP-conjugated affinity-purified goat anti-rabbit IgG, were purchased from Sigma-Aldrich. Protein Extraction and Quantitation Kits were purchased from Bio-Rad Laboratories, Inc. GEM was purchased from Eli Lilly and Company (USA and Canada).
Construction of the recombinant HMGB1 expression vector
The human HMGB1 mRNA sequence was acquired from GenBank (NM_002128.4 in GenBank). Primer design using the flank of the HMGB1 ORF and the restriction enzyme analysis were performed by Primer Premier five software.
Total RNA was extracted from PANC-1 cells and then quantified according to the manufacturer’s instructions of the Trizol reagent. The first strand of cDNA was synthesized from the mRNA template from the PANC-1 cells, and the PCR was conducted with the HMGB1 primers. The PCR products were cloned into the pGEM-T vector. After cleavage and identification with restriction endonucleases, the correct recombinant plasmid was sequenced. The pcDNA3.1 vector and the pGEM-HMGB1 recombinant plasmid were simultaneously cleaved with the BamH I and EcoR I restriction endonucleases, and the target fragments were joined by T4 DNA ligase. Finally, the pcDNA3.1-HMGB1 recombinant plasmid was transformed into DH5α competent cells.
Cell treatment
BxPC-3 cells were cultured in RPMI 1640 medium containing 10% FBS, 10 mM HEPES, 1.5 g/L NaHCO3 and 2 mM L-glutamine. PANC-1 cells were cultured in DMEM supplemented with 10% FBS, 1.5 g/L NaHCO3 and 4 mM L-glutamine. Both BxPC-3 and PANC-1 cells were cultured under standard conditions (37 °C, 5% CO2 and saturated humidity). The growth state of the cells was observed under an inverted microscope. Once the cells were at 70% to 80% confluency, cells were digested with 0.25% trypsin and passaged. The cells were passaged every 3 to 4 days, and the culture medium was changed every other day. Cells in logarithmic growth phase were harvested for future assays.
Cultured PANC-1 cells were seeded uniformly into six-well culture plates at a density of 3×105 cells/mL. The volume of cells in each well was 1,000 µL. After the cells adhered to the culture surface, the miR-218 mimic, non-specific control (mimic ctrl), recombinant HMGB1 expression vector (pcDNA3.1-HMGB1) and empty pcDNA3.1 vector (vector ctrl) were transfected into the cells using the Lipofectamine 2000 transfection reagent in accordance with the manufacturer’s instructions. The normal control group (normal ctrl) was established at the same time. The ciR-218 mimic, mimic ctrl, pcDNA3.1-HMGB1 and vector ctrl were diluted in serum-free Minimum Essential Media (MEM). Subsequently, the Lipofectamine 2000 liposome was mixed gently with MEM and incubated at room temperature (RT) for 5 min. The MEM-diluted Lipofectamine 2000 was then mixed with each of the miR-218 mimic, mimic ctrl, pcDNA3.1-HMGB1 and vector ctrl. The mixtures were incubated at RT for 20 min to allow formation of complexes. The complexes were added to the culture plates containing PANC-1 cells and mixed gently. The cells were then placed into a 37 °C and 5% CO2 (volume fraction) incubator. After 5 h of incubation, the complex-containing medium was replaced with either fresh MEM supplemented with 10% FBS or MEM containing 10% FBS and 5 µM GEM (final concentration). The cells were cultured for an additional 48 h.
Examination of the effect of GEM on miR-218 expression in human pancreatic cancer cell lines
We first examined the differences in miR-218 expression between GEM-sensitive BxPC-3 pancreatic cancer cells and GEM-resistant PANC-1 cells using quantitative reverse transcription polymerase chain reaction (qRT-PCR). In vitro cultured BxPC-3 and PANC-1 pancreatic cancer cells were harvested. RNA was extracted from the cells using the TaqMan miRNA Isolation Kit. The expression of mature miR-218 was examined using the TaqMan microRNA Assay and TaqMan Universal PCR Master Mix. The U6 small nuclear RNA (snRNA) gene was used as an internal reference gene. Three replica wells were set up for each reaction. The threshold cycle (CT) value of the sample in each reaction tube was recorded, and the qRT-PCR results were analyzed using the relative quantification method.
The effect of GEM on miR-218 expression in human pancreatic cancer cell lines was examined by qRT-PCR. PANC-1 cells were seeded uniformly into six-well culture plates at a density of 3×105 cells/mL. One group of PANC-1 cells was treated with GEM at a final concentration of 5 µM for 48 h. The normal ctrl group was set up at the same time. These two groups of cells were collected, and the RNA was extracted to measure the expression levels of mature miR-218. Three replica wells were set up for each reaction. The CT values of the samples in all reaction tubes were recorded, and the qRT-PCR results were analyzed using the relative quantification method.
Examination of the effect of miR-218 on the viability of GEM-induced pancreatic cancer cells using the CCK-8 assay
To investigate the effect of miR-218 on the viability of GEM-induced pancreatic cancer cells, miR-218 was overexpressed in PANC-1 cells by transfection of a miR-218 mimic, and the mimic ctrl and normal ctrl groups were set up at the same time. At 48 h after transfection, RNA was extracted from all groups using the TaqMan miRNA Isolation Kit, and changes in miR-218 expression among the groups were analyzed using qRT-PCR.
Cell viability was examined using the CCK-8 Assay Kit according to the manufacturer’s instructions. PANC-1 cells were transfected with the miR-218 mimic or mimic ctrl. Subsequently, GEM was added to each well at a final concentration of 5 µM. After treatment of the cells with GEM for 48 h, the CCK-8 solution was added to the cells, and the cells were incubated at 37 °C in a 5% CO2 incubator for 2 h. The normal ctrl group was set up at the same time. The optical density (OD) of the cells was measured at 450 nm (reference wavelength of 630 nm) using a microplate reader (Bio-Tek, USA). Ten replica wells were set up for each experimental group, and each experiment was repeated three times.
Examination of the effect of miR-218 on the apoptosis of GEM-induced cells by flow cytometric analysis
To verify the effect of miR-218 on the drug sensitivity of GEM-induced pancreatic cancer cells, flow cytometric analysis was performed to examine the effect of miR-218 on GEM-induced apoptosis. PANC-1 pancreatic cancer cells were seeded uniformly into six-well culture plates at a density of 3×105 cells/mL. After PANC-1 cells were transfected with the miR-218 mimic or mimic ctrl, GEM was added to each well at a final concentration of 5 µM for 48 h. The normal ctrl group was established at the same time. Subsequently, the cells were washed 1-2 times with PBS and labeled with Annexin V-Fluorescein isothiocyanate (FITC) and PI for 15 min at room temperature (RT) in the dark. The cells were then filtered through cell sieves and examined by flow cytometric analysis (BD Biosciences, USA). The number of cells was counted using FCM CellQuest software, and the data were analyzed using Macquit software.
Examination of the effect of GEM on HMGB1 expression in pancreatic cancer cells by western blot analysis
Previous studies have demonstrated that HMGB1 is related to the sensitivity of tumors to chemotherapy drugs (27,28). In addition, our preliminary results showed that HMGB1 is a target gene of miR-218. miR-218 affects the biological characteristics of pancreatic cancer cells through regulating the expression of HMGB1. We postulated that miR-218 affects the sensitivity of pancreatic cancer cells to GEM through regulating HMGB1. Therefore, we conducted western blot analysis to examine whether GEM affects the expression of HMGB1 in pancreatic cancer cells.
PANC-1 pancreatic cancer cells were seeded uniformly into six-well culture plates at a density of 3×105 cells/mL. One group of PANC-1 cells was treated with GEM (final concentration of 5 µM) for 48 h. Subsequently, 1 mL of radioimmunoprecipitation assay (RIPA) lysis buffer [150 mM NaCl, 1% NP40, 0.5% sodium deoxycholate, 0.1% SDS, 50 mM Tris (pH 7.9), 10 mM NaF, 10 mM PMSF and 1× protease inhibitors (Complete cocktail tablets, Roche)] was added to each well. The cell lysates were transferred to 1.5 mL centrifuge tubes and centrifuged at 16,000 g for 30 min, and the supernatants were then collected. The protein concentrations were measured using the bicinchoninic acid (BCA) assay, and 50 µg of total protein was loaded in each lane of a gel composed of a 5% stacking gel and 15% separating gel. The proteins were separated by electrophoresis and transferred to a polyvinylidene difluoride (PVDF) membrane using a wet transfer system (Bio-Rad Laboratories, Inc., USA). The membrane was blocked at RT in Tris-Buffered Saline-Tween 20 (TBST) solution (10 mM Tris-HCl, pH 7.5; 150 mM NaCl; and 0.1% Tween-20) containing 5% nonfat dry milk for 1 h and then incubated with the rabbit anti-human HMGB1 monoclonal antibody (1:500 dilution) and mouse anti-human β-actin monoclonal antibody (1:1,000 dilution) at 4 °C overnight. The membrane was washed three times (5 min each wash) with TBST and incubated with HRP-labeled goat anti-rabbit IgG or HRP-labeled goat anti-mouse IgG secondary antibody at 37 °C for 1 h. The membrane was washed three times (5 min each wash) with TBST, incubated with enhanced chemiluminescence (ECL) reagent and autoradiographed. The relative content of HMGB1 is expressed as the grayscale ratio of HMGB1/β-actin, and the changes in the relative HMGB1 expression were analyzed using PDQuest software (Bio-Rad Laboratories, Inc., Richmond, CA).
Investigation of the effects of miR-218 on the sensitivity of pancreatic cancer cells to GEM through regulation of HMGB1
To further verify the mechanism by which miR-218 affects the drug sensitivity of GEM-induced pancreatic cancer cells, HMGB1 was ectopically expressed in PANC-1 pancreatic cancer cells to investigate whether HMGB1 expression alters the effect of miR-218 on the sensitivity of the cells to GEM. To achieve the ectopic expression of HMGB1, an HMGB1 recombinant expression vector was constructed. Subsequently, HMGB1 siRNA or the recombinant HMGB1 expression vector (pcDNA3.1-HMGB1) was transfected into PANC-1 pancreatic cancer cells, and the normal ctrl group was set up at the same time. At 48 h after transfection, proteins were extracted from each group, and western blot analysis was performed to examine the expression of HMGB1.
To investigate whether miR-218 affects the sensitivity of pancreatic cancer cells to GEM through regulation of HMGB1, flow cytometric analysis was performed. The cells were divided into the following three groups: the pcDNA3.1-HMGB1 transfected group; the miR-218 mimic and pcDNA3.1-HMGB1 cotransfected group (miR-218 mimic + pcDNA3.1-HMGB1); and the miR-218 mimic and HMGB1 siRNA cotransfected group (miR-218 mimic + HMGB1 siRNA). All three groups were treated with GEM (final concentration of 5 µM) for 48 h. The cells were then washed one to two times with PBS and labeled with Annexin V-FITC and PI dye for 15 min at RT in the dark. After sieve filtration, the cells were subjected to flow cytometric analysis (BD Biosciences, USA). The number of cells was counted using FCM CellQuest software, and the data were analyzed using Macquit software.
Statistical analysis
Experimental data were statistically analyzed using SPSS17.0 statistical analysis software. Comparison between two groups was conducted using the t-test, while comparisons between multiple groups of data were performed using analysis of variance (ANOVA). P values less than 0.05 were considered statistically significant.
Results
The effect of GEM on miR-218 expression in human pancreatic cancer cell lines
qRT-PCR was performed to examine the difference in miR-218 expression between GEM-sensitive BxPC-3 pancreatic cancer cells and GEM-resistance PANC-1 cells. The qRT-PCR results showed that the expression of miR-218 was significantly lower in GEM-resistant PANC-1 cells than in GEM-sensitive BxPC-3 cells (P<0.05) (Figure 1A).
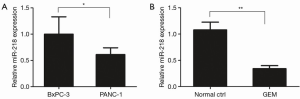
In addition, qRT-PCR was performed to investigate the effect of GEM on miR-218 expression in the PANC-1 human pancreatic cancer cell line. Compared with the normal ctrl group, the miR-218 expression was markedly reduced in PANC-1 cells after treatment with 5 µM GEM for 48 h (P<0.01) (Figure 1B).
The effect of miR-218 on the viability of GEM-induced pancreatic cancer cells by the CCK-8 assay
miR-218 was overexpressed via transfection of the miR-218 mimic in PANC-1 cells, and the effect of miR-218 overexpression on miR-218 expression was examined using qRT-PCR. The qRT-PCR results showed that the miR-218 expression was significantly increased in the miR-218 mimic-transfected group compared to the normal ctrl group (P<0.01) and the negative control (mimic ctrl) group (P<0.01) (Figure 2A).
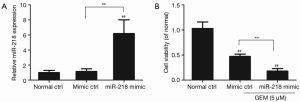
The effect of miR-218 overexpression on the viability of GEM-induced pancreatic cancer cells was analyzed using CCK-8. PANC-1 cells were transfected with the miR-218 mimic or mimic control (mimic ctrl), treated with 5 µM GEM for 48 h, and subjected to the CCK-8 assay. Figure 2B shows that cell viability was significantly lower in the miR-218 mimic-transfected group or in the mimic ctrl group in comparison with the normal ctrl group (P<0.01). In addition, cell viability was markedly decreased in the miR-218 mimic-transfected group compared to the negative control (mimic ctrl) group (P<0.01) (Figure 2B). These results indicated that treatment of PANC-1 cells with 5 µM GEM significantly reduced cell viability and that miR-218 enhanced the sensitivity of PANC-1 cells to GEM.
The effect of miR-218 on the apoptosis of GEM-induced cells by a flow cytometric assay
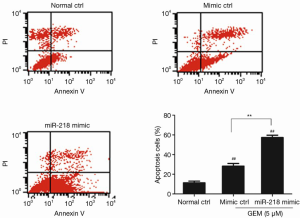
PANC-1 cells were transfected with the miR-218 mimic or mimic ctrl, treated with 5 µM GEM for 48 h, and then subjected to flow cytometric analysis. The results showed that the percentage of apoptotic cells was significantly higher in the miR-218 mimic-transfected group and the mimic ctrl group in comparison to the normal ctrl group (P<0.01) (Figure 3). Moreover, the percentage of apoptotic cells was significantly increased in the miR-218 mimic-transfected group compared with the negative control (mimic ctrl) group (P<0.01). The above results indicated that treatment of PANC-1 cells with 5 µM GEM significantly increased the proportion of apoptotic cells and that miR-218 promoted the apoptosis of GEM-induced pancreatic cancer cells.
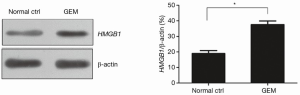
The effect of GEM on HMGB1 expression in pancreatic cancer cells by western blot analysis
The results of western blot analysis showed that the expression level of HMGB1 was significantly increased in PANC-1 cells after treatment with 5 µM GEM for 48 h (P<0.05) (Figure 4). These results demonstrated that GEM induced the expression of HMGB1 in PANC-1 cells.
The miR-218 effect on the sensitivity of pancreatic cancer cells to GEM through regulation of HMGB1
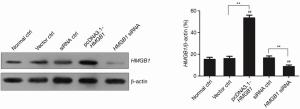
PANC-1 pancreatic cancer cells were transfected with HMGB1 siRNA or the recombinant HMGB1 expression vector (pcDNA3.1-HMGB1). The changes in HMGB1 expression in PANC-1 cells were examined by western blot analysis. Figure 5 shows that the HMGB1 expression level was significantly increased in the pcDNA3.1-HMGB1 transfected group compared with the normal ctrl group (P<0.01) and that the HMGB1 expression level was significantly reduced in the HMGB1 siRNA transfected group (P<0.01). These results indicated that both HMGB1 siRNA and the recombinant HMGB1 expression vector (pcDNA3.1-HMGB1) exerted potent effects in PANC-1 cells.
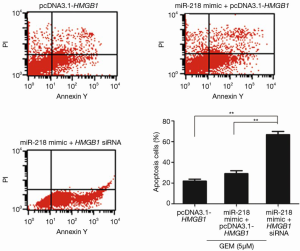
Flow cytometric analysis was performed to examine whether miR-218 affects the sensitivity of pancreatic cancer cells to GEM through the regulation of HMGB1. The results showed that approximately 21.98% PANC-1 cells underwent apoptosis after the cells were transfected with pcDNA3.1-HMGB1 and treated with 5 µM GEM (Figure 6), and the results also showed that the percentage of apoptotic PANC-1 cells was approximately 25.61% when the cells were treated with 5 µM GEM alone (Figure 3). These results demonstrated that HMGB1 inhibited the apoptosis of GEM-induced PANC-1 cells. The percentage of apoptotic PANC-1 cells was approximately 27.35% when the cells were cotransfected with the miR-218 mimic and pcDNA3.1-HMGB1 followed by treatment with 5 µM GEM (the miR-218 mimic + pcDNA3.1-HMGB1 + GEM group) (Figure 6), which was significantly lower in comparison to the cells transfected with the miR-218 mimic and treated with 5 µM GEM (the miR-218 mimic + GEM group). These results indicated that HMGB1 reversed the effect of miR-218 on the drug sensitivity of GEM-induced PANC-1 cells. In contrast, the percentage of apoptotic cells in the miR-218 mimic and HMGB1 siRNA cotransfected group (the miR-218 mimic + HMGB1 siRNA group) reached 62.67% (Figure 6). These results indicated that the reduction of HMGB1 levels enhanced the effect of miR-218 on the drug sensitivity of GEM-induced PANC-1 cells.
Discussion
Pancreatic cancer is one of the most common gastrointestinal tract malignancies. Pancreatic cancer exhibits an extremely high degree of malignancy. Due to its deep anatomical location within the abdomen and insidious onset, early diagnosis of pancreatic cancer is rather difficult. Most cases of pancreatic cancer are discovered at advanced stages. Moreover, pancreatic cancer exhibits limited sensitivity to radiochemotherapy, and surgical resection is difficult to achieve. Therefore, pancreatic cancer typically shows poor prognosis and has become one of the cancer types that pose the greatest threat to human health (29,30). Chemotherapy is an important adjunctive treatment for pancreatic cancer and plays a role in improving the quality of life of patients and prolonging patient survival (31,32). Currently, GEM is a first-line drug used in chemotherapeutic treatment of pancreatic cancer. However, the overall efficacy of GEM is less than 20% (33,34). The main problem of GEM treatment is that the majority of pancreatic cancer patients will develop resistance to GEM (35). Therefore, identification of genes related to GEM resistance in pancreatic cancer and enhancement of GEM efficacy are the issues that urgently need solutions in clinical practices.
Studies have shown that the failure of chemotherapy in the treatment of pancreatic cancer is attributed mainly to chemoresistance. Factors capable of inducing chemoresistance in tumors are multifaceted, including intracellular drug sequestration, drug efflux, metabolic detoxification, alteration of the molecular targets of the drugs, enhancement of DNA repair capacity and regulation of apoptosis (36,37). Currently, extensive research is being conducted at the gene and protein levels to investigate the mechanism underlying the development of chemoresistance in tumors. A number of chemoresistance-related genes have been identified, including P-glycoprotein (P-gp), multidrug resistance protein (MRP), glutathione S-transferase π (GST-π) and topoisomerase II (TopoII) (38). However, no significant reduction in tumor chemoresistance has been observed clinically after intervention, specifically with the above genes and their upstream/downstream targets (39). Therefore, in-depth studies of a wider range of chemoresistance-related factors are of great significance.
Studies on the relationship between miRNAs and the chemosensitivity of tumors have received increasing attention from scholars in the field of oncology and have become a research hotspot in recent years. Our previous study has shown that miR-218 expression is downregulated in human pancreatic cancer tissues and cell lines. miR-218 negatively regulates the expression of HMGB1 protein and inhibits the proliferation and invasion of pancreatic cancer cells. Previous studies have confirmed that miR-218 is a tumor suppressing microRNA. A study conducted by Li et al. has shown that miR-218 inhibits the growth of cervical cancer and promotes the sensitivity of cervical cancer to cisplatin (26). However, the relationship between miR-218 and the sensitivity of pancreatic cancer cells to GEM-based chemotherapy has not been reported. In the present study, we performed qRT-PCR to examine the differences in miR-218 expression between GEM-sensitive BxPC-3 pancreatic cancer cells and GEM-resistant PANC-1 cells. The results showed that miR-218 expression was significantly lower in GEM-resistant PANC-1 cells compared to GEM sensitive BxPC-3 cells (P<0.05), thus indicating that the resistance of PANC-1 cells to GEM might be related to miR-218. To further explore the correlation between miR-218 and the resistance of PANC-1 cells to GEM, the CCK-8 assay and flow cytometric analysis were conducted to examine the effects of miR-218 on the viability and apoptosis of GEM-induced pancreatic cancer cells. The results demonstrated that treatment of PANC-1 cells with 5 µM GEM significantly reduced cell viability and promoted apoptosis. The results also demonstrated that miR-218 enhanced the sensitivity of PANC-1 cells to GEM.
HMGBl is member of the high mobility group (HMG) protein family. HMGB1 is abundantly expressed in the nuclei of eukaryotic cells (40,41). Long-term studies have suggested that HMGB1 is a typical non-histone chromosomal protein. Through binding to DNA, HMGB1 participates in the construction and maintenance of nucleosomes and plays various roles in the processes of transcription, replication, recombination, DNA repair and genomic stability maintenance (42,43). To date, studies have found that HMGB1 can be actively secreted by inflammatory cells and (or) tumor cells as well as passively released into the extracellular environment from necrotic cells. As an inflammatory factor, HMGB1 binds to multiple cell surface receptors, primarily to the receptor for advanced glycation end products (RAGE), which activates a series of downstream signaling pathways. Therefore, HMGB1 plays a crucial role in inflammation, cancer, sepsis, autoimmune diseases and vascular lesions (44,45). Previous studies have reported that HMGB1 is related to the chemosensitivity of tumor cells (46,47). Angelova et al. further confirmed that HMGB1 is related to the sensitivity of pancreatic cancer cells to GEM-based chemotherapy (48).
Our previous study confirmed that HMGB1 is a target gene of miR-218. miR-218 affects the biological characteristics of pancreatic cancer cells through regulating the expression of HMGB1. To further verify the mechanism by which miR-218 affects the drug sensitivity of GEM-induced pancreatic cancer cells, HMGB1 was ectopically expressed in PANC-1 pancreatic cancer cells to examine whether HMGB1 expression modulates the effect of miR-218 on the sensitivity of PANC-1 cells to GEM. The results of flow cytometric analysis showed that overexpression of HMGB1 in PANC-1 pancreatic cancer cells reversed the effect of miR-218 on the drug sensitivity in GEM-induced PANC-1 cells. These results indicated that miR-218 affected the sensitivity of pancreatic cancer cells to GEM through regulating the expression of HMGB1.
Currently, the mechanisms underlying GEM resistance have become a hotspot in the field of pancreatic cancer research worldwide. Understanding the mechanisms of GEM resistance in pancreatic cancer is conducive to predicting the resistance of pancreatic cancer to GEM and to providing better treatment for pancreatic cancer (33,49). Various studies on the mechanisms underlying GEM resistance in pancreatic cancer have provided different discoveries and insights (5,50). In short, the resistance of pancreatic cancer cells to GEM results from the joint effect of multiple elements, multiple factors and multiple pathways. Any single mechanism is unable to fully explain the phenomenon of GEM resistance. Therefore, the mechanism by which miR-218 affects the drug sensitivity in GEM-induced PANC-1 cells is still unclear and needs further investigation.
In summary, miR-218 expression is downregulated in the GEM-resistant cell line, PANC-1. Overexpression of miR-218 enhances the sensitivity of PANC-1 cells to GEM. The mechanism of the miR-218 effect may be through the regulation of HMGB1 expression.
Acknowledgements
Funding: This work was supported by grants: (I) Liaoning Provincial Department of Education Science Research Project (L2014299); (II) Liaoning Province Science and Technology Plan Project (2011404013-4); (III) The Shenyang Municipal Science and Technology Project (F12-277-1-73).
Disclosure: The authors declare no conflict of interest.
References
- Singh HM, Ungerechts G, Tsimberidou AM. Gene and cell therapy for pancreatic cancer. Expert Opin Biol Ther 2015;15:505-16. [PubMed]
- Zhang C, Yang G, Ling Y, et al. The early diagnosis of pancreatic cancer and diabetes: what's the relationship? J Gastrointest Oncol 2014;5:481-8. [PubMed]
- Chen W, Zheng R, Zhang S, et al. Report of cancer incidence and mortality in China, 2010. Ann Transl Med 2014;2:61. [PubMed]
- Ito M, Makino N, Ueno Y. Glucose intolerance and the risk of pancreatic cancer. Transl Gastrointest Cancer 2013;2:223-9.
- Furuse J, Nagashima F. Current status and future direction of chemotherapy for pancreatic cancer. Chin Clin Oncol 2013;2:6. [PubMed]
- Xu K, Niu L, Yang D. Cryosurgery for pancreatic cancer. Gland Surg 2013;2:30-9. [PubMed]
- Zaccagna F, Anzidei M, Sandolo F, et al. MRgFUS for liver and pancreas cancer treatments: the Umberto I hospital experience. Transl Cancer Res 2014;3:430-41.
- Palmieri G, Buonerba C, Ottaviano M, et al. Capecitabine plus gemcitabine in thymic epithelial tumors: final analysis of a Phase II trial. Future Oncol 2014;10:2141-7. [PubMed]
- Francis T, Graf A, Hodges K, et al. Histamine regulation of pancreatitis and pancreatic cancer: a review of recent findings. Hepatobiliary Surg Nutr 2013;2:216-26. [PubMed]
- Fiteni F, Nguyen T, Vernerey D, et al. Cisplatin/gemcitabine or oxaliplatin/gemcitabine in the treatment of advanced biliary tract cancer: a systematic review. Cancer Med 2014;3:1502-11. [PubMed]
- Delavan JA, Chino JP, Vinson EN. Gemcitabine-induced radiation recall myositis. Skeletal Radiol 2015;44:451-5. [PubMed]
- Blanco FF, Pishvaian MJ, Brody JR. Upgrading gemcitabine with recycled kinase inhibitors. Cell Cycle 2014;13:2810-1. [PubMed]
- Li Q, Yuan Z, Yan H, et al. Comparison of gemcitabine combined with targeted agent therapy versus gemcitabine monotherapy in the management of advanced pancreatic cancer. Clin Ther 2014;36:1054-63. [PubMed]
- Lee HW, Chung MJ, Kang H, et al. Gemcitabine-induced hemolytic uremic syndrome in pancreatic cancer: a case report and review of the literature. Gut Liver 2014;8:109-12. [PubMed]
- Gupta AA, Yao X, Verma S, et al. Chemotherapy (gemcitabine, docetaxel plus gemcitabine, doxorubicin, or trabectedin) in inoperable, locally advanced, recurrent, or metastatic uterine leiomyosarcoma: a clinical practice guideline. Curr Oncol 2013;20:e448-54. [PubMed]
- Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 2013;369:1691-703. [PubMed]
- Kuroda T, Kumagi T, Yokota T, et al. Improvement of long-term outcomes in pancreatic cancer and its associated factors within the gemcitabine era: a collaborative retrospective multicenter clinical review of 1,082 patients. BMC Gastroenterol 2013;13:134. [PubMed]
- Jordheim LP, Dumontet C., Do . hENT1 and RRM1 predict the clinical benefit of gemcitabine in pancreatic cancer? Biomark Med 2013;7:663-71. [PubMed]
- Heinemann V, Haas M, Boeck S. Neoadjuvant treatment of borderline resectable and non-resectable pancreatic cancer. Ann Oncol 2013;24:2484-92. [PubMed]
- Bhattacharjee V, Zhou Y, Yen TJ. A synthetic lethal screen identifies the Vitamin D receptor as a novel gemcitabine sensitizer in pancreatic cancer cells. Cell Cycle 2014;13:3839-56. [PubMed]
- Wang Q, Wei L, Guan X, et al. Briefing in family characteristics of microRNAs and their applications in cancer research. Biochim Biophys Acta 2014;1844:191-7.
- Kaplan BB, Kar AN, Gioio AE, et al. MicroRNAs in the axon and presynaptic nerve terminal. Front Cell Neurosci 2013;7:126. [PubMed]
- Li M, Fu W, Wo L, et al. miR-128 and its target genes in tumorigenesis and metastasis. Exp Cell Res 2013;319:3059-64. [PubMed]
- Leite-Moreira AM, Lourenço AP, Falcão-Pires I, et al. Pivotal role of microRNAs in cardiac physiology and heart failure. Drug Discov Today 2013;18:1243-9. [PubMed]
- Piva R, Spandidos DA, Gambari R. From microRNA functions to microRNA therapeutics: novel targets and novel drugs in breast cancer research and treatment Int J Oncol 2013;43:985-94. [PubMed]
- Li J, Ping Z, Ning H. MiR-218 impairs tumor growth and increases chemo-sensitivity to cisplatin in cervical cancer. Int J Mol Sci 2012;13:16053-64. [PubMed]
- Amornsupak K, Insawang T, Thuwajit P, et al. Cancer-associated fibroblasts induce high mobility group box 1 and contribute to resistance to doxorubicin in breast cancer cells. BMC Cancer 2014;14:955. [PubMed]
- Chen GY, Meng CL, Lin KC, et al. Graphene oxide as a chemosensitizer: diverted autophagic flux, enhanced nuclear import, elevated necrosis and improved antitumor effects. Biomaterials 2015;40:12-22. [PubMed]
- Zarogoulidis P, Pavlioglou P, Pivert PL, et al. Current and future intratumoral targeted treatment for pancreatic cancer. Ther Deliv 2014;5:913-26. [PubMed]
- Li X, Truty MA, Kang Y, et al. Extracellular lumican inhibits pancreatic cancer cell growth and is associated with prolonged survival after surgery. Clin Cancer Res 2014;20:6529-40. [PubMed]
- Fuchs CS, Azevedo S, Okusaka T, et al. A phase 3 randomized, double-blind, placebo-controlled trial of ganitumab or placebo in combination with gemcitabine as first-line therapy for metastatic adenocarcinoma of the pancreas: the GAMMA trial†. Ann Oncol 2015;26:921-7. [PubMed]
- Watanabe T, Ueno H, Watabe Y, et al. ACTN4 copy number increase as a predictive biomarker for chemoradiotherapy of locally advanced pancreatic cancer. Br J Cancer 2015;112:704-13. [PubMed]
- Fiorini C, Cordani M, Gotte G, et al. Onconase induces autophagy sensitizing pancreatic cancer cells to gemcitabine and activates Akt/mTOR pathway in a ROS-dependent manner. Biochim Biophys Acta 2015;1854:549-60.
- Sun M, Zhao W, Xie Q, et al. Lentinan reduces tumor progression by enhancing gemcitabine chemotherapy in urothelial bladder cancer. Surg Oncol 2015;24:28-34. [PubMed]
- Sherman WH, Chu K, Chabot J, et al. Neoadjuvant gemcitabine, docetaxel, and capecitabine followed by gemcitabine and capecitabine/radiation therapy and surgery in locally advanced, unresectable pancreatic adenocarcinoma. Cancer 2015;121:673-80. [PubMed]
- Jagadeesh D, Smith MR. Novel targeted therapies in peripheral T cell lymphoma. Discov Med 2013;15:367-78. [PubMed]
- Morotti M, Becker CM, Menada MV, et al. Targeting tyrosine-kinases in ovarian cancer. Expert Opin Investig Drugs 2013;22:1265-79. [PubMed]
- Lubin J, Markowska A, Knapp P. Factors affecting response of chemotherapy in women with ovarian cancer. Eur J Gynaecol Oncol 2012;33:644-7. [PubMed]
- Breier A, Gibalova L, Seres M, et al. New insight into p-glycoprotein as a drug target. Anticancer Agents Med Chem 2013;13:159-70. [PubMed]
- Srinivasan M, Banerjee S, Palmer A, et al. HMGB1 in hormone-related cancer: a potential therapeutic target. Horm Cancer 2014;5:127-39. [PubMed]
- Pisetsky DS. The expression of HMGB1 on microparticles released during cell activation and cell death in vitro and in vivo. Mol Med 2014;20:158-63. [PubMed]
- Andersson U, Antoine DJ, Tracey KJ. The functions of HMGB1 depend on molecular localization and post-translational modifications. J Intern Med 2014;276:420-4. [PubMed]
- Li LC, Gao J, Li J. Emerging role of HMGB1 in fibrotic diseases. J Cell Mol Med 2014;18:2331-9. [PubMed]
- Lu B, Wang C, Wang M, et al. Molecular mechanism and therapeutic modulation of high mobility group box 1 release and action: an updated review. Expert Rev Clin Immunol 2014;10:713-27. [PubMed]
- Huebener P, Pradere JP, Hernandez C, et al. The HMGB1/RAGE axis triggers neutrophil-mediated injury amplification following necrosis. J Clin Invest 2015;125:539-50. [PubMed]
- Cottone L, Capobianco A, Gualteroni C, et al. 5-Fluorouracil causes leukocytes attraction in the peritoneal cavity by activating autophagy and HMGB1 release in colon carcinoma cells. Int J Cancer 2015;136:1381-9. [PubMed]
- Pan B, Chen D, Huang J, et al. HMGB1-mediated autophagy promotes docetaxel resistance in human lung adenocarcinoma. Mol Cancer 2014;13:165. [PubMed]
- Angelova AL, Grekova SP, Heller A, et al. Complementary induction of immunogenic cell death by oncolytic parvovirus H-1PV and gemcitabine in pancreatic cancer. J Virol 2014;88:5263-76. [PubMed]
- Yang MH, Lee KT, Yang S, et al. KML001 enhances anticancer activity of gemcitabine against pancreatic cancer cells. Anticancer Res 2015;35:183-9. [PubMed]
- Ju HQ, Gocho T, Aguilar M, et al. Mechanisms of Overcoming Intrinsic Resistance to Gemcitabine in Pancreatic Ductal Adenocarcinoma through the Redox Modulation. Mol Cancer Ther 2015;14:788-98. [PubMed]


