Th22 cells are associated with hepatocellular carcinoma development and progression
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignancies and the third most frequent cause of cancer death in the world (1). The pathogenesis of HCC is heterogeneous and complex. Many evidences demonstrate that immune response plays a critical role in the pathogenesis, invasion and metastasis of HCC (2). It has been well recognized that the adaptive immune response by CD4+ T cells plays an important role in controlling the development of the malignancy. To date, most studies on tumor immunity in HCC mainly focus on Th1/Th2. Recently, a novel subset of CD4+ T cells, IL-22-producing CD4+ T helper cells (Th22 cells), have been identified and showed to challenge the classical Th1/Th2 paradigm (3,4).
Th22 cells are inflammatory CD4+ T cells that secret IL-22 but do not express IL-17 or interferon-gamma (IFN-γ) (5-7). Th22 cells are distinct in the profile of altering genes compared with other T cells, such as Th1, Th2 and Th17, indicating that Th22 cells have an individual signature (7). The mainly effector cytokine of Th22 cells is IL-22, which belongs to the IL-10 cytokine family. IL-22 mediates its effects via a heterodimeric transmembrane receptor complex consisting of IL-22R1 and IL-10R2, and subsequently activates Janus kinase and signal transducer and activator of transcription (JAK-STAT) signaling pathways, including STAT-3, Jak1 and Tyk2 (8).
Recent studies have implicated the role of Th22 cells and their effector cytokine, IL-22, in the pathogenesis of several autoimmune diseases and tumors in humans, such as Crohn’s disease (9), gastric cancer (10) and lung cancer (11). To date, no previous study has reported data regarding the role of Th22 cells in the pathogenesis of HCC. Therefore, the present study aimed to determine the levels of Th22 cells and IL-22 in HCC patients.
Materials and methods
Subjects and samples
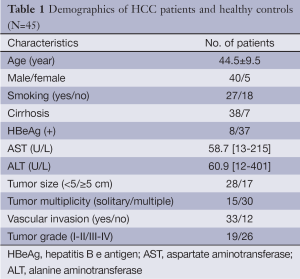
Forty-five newly diagnosed HCC patients were recruited from the First Affiliated Hospital of Guangxi Medical University between October 2012 and June 2013. The diagnosis of HCC was based on pathology test, serology test and radiology test. The fresh HCC tissues, adjacent HCC tissues as well as peripheral blood samples were collected. Adjacent HCC tissue is defined as tissue that locates within 1 cm of the HCC and confirmed to be nontumor tissue by pathology. Peripheral blood samples from 19 healthy controls and 10 normal liver tissues from other liver surgery were also collected. The study was approved by the Review Boards of the Guangxi Medical University. Written informed consent was obtained from each participant. The demographics of the HCC and healthy controls enrolled are shown in Table 1.
Full table
HCC patients with autoimmune diseases and other types of carcinoma were excluded. None of the patients had received anticancer treatment prior to surgery. Serum was separated from the blood by centrifugation at 2,500 r/min for 10 min at 4 °C, and stored at –80 °C for later analysis with enzyme-linked immunosorbent assay (ELISA) and flow cytometry analysis. After surgical removal, the fresh liver tissue was divided into two parts. One part was fixed in 4% paraformaldehyde solution and later embedded in paraffin for hematoxylin-eosin (H&E) and immunohistochemical staining. The other part was immediately frozen in liquid nitrogen for future real-time polymerase chain reaction (PCR).
Flow cytometry analysis
The following antibodies were used to stain single-cell suspensions: CD3-APC-H7, IL-17-PE, IFN-γ-PE-Cy7 (BD Pharmingen, San Diego, CA, USA), CD4-FITC, IL-22-Alexa Flu- or 647, CD25-PE, and Foxp3-APC (eBioscience, San Diego, CA, USA). Peripheral blood was collected in heparinized tubes. Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll-Paque density gradient centrifugation. PBMCs were resuspended at 1×106 cells/mL in RPMI 1640 containing 10% fetal calf serum (FCS) and 100 U/mL penicillin-streptomycin. The cells were then stimulated for 4 h with phorbol 12-myristate 13-acetate (PMA; 50 ng/mL; Sigma-Aldrich, St. Louis, MO, USA) and ionomycin (1 µg/mL; Sigma-Aldrich) in the presence of Golgistop reagent (0.7 µL/mL, BD Biosciences, San Diego, CA, USA). Stimulated or not stimulated PBMCs were washed twice with PBS containing 1% FCS and incubated with surface antibodies for 30 min at 4 °C, and then fixed and permeabilized for 20 min using Cytofix/Cytoperm reagent (BD Biosciences, USA). The cells were then washed and stained with antibodies to intracellular molecules. Isotype antibodies were used as controls. Cells were collected by flow cytometry with a FACSCanto II (BD Biosciences, USA). Data were analyzed with Flowjo software (TreeStar) or FACS-Diva software (BD Biosciences, USA).
Serum IL-22 level determination
Serum IL-22 levels were determined using ELISA kits (R&D Systems) according to the manufacturer’s instructions.
Immunohistochemical staining for IL-22
Paraffin sections (4 µm) were stained with rabbit polyclonal antibody for IL-22 (rabbit polyclonal, 1:200, Santa Cruz Biotechnology, Santa Cruz, California, USA). Streptavidin peroxidase (SP) method was performed according to the manufacturer’s instructions. The number of positive cells was counted under high power (HP) magnification (×400), and ten HP fields were randomly selected and scored by two independent observers who were unaware of the diagnosis and clinical data.
Quantitative real-time PCR analysis
Total RNA was isolated by Trizol (Invitrogen, America) according to the manufacturer’s instructions. Approximately, 1 µg of total RNA from each sample was subjected to first-strand cDNA synthesis using RevertAidTM First Strand cDNA Synthesis Kit (MBI, Fermentas, USA). Reverse transcription reaction was done at 42 °C for 1 h, followed by 95 °C for 5 min. Real-time PCR was conducted using Applied Biosystems 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) in accordance to the manufacturer’s instructions. The real-time PCR contained, in a final volume of 20 µL, 10 µL of 2× SYBR Green Real-time PCR Master Mix, 1 µL of cDNA, and 1 µL of the forward and reverse primers. The primers for human IL-22 and β-actin were as follows: IL-22 forward, 5'-ACT GGA TTT GCT GTT TAT GTC TCT G-3' and reverse, 5'-GGC TTC CCA TCT TCC TTT TG-3'; IL-17 forward, 5'-CTC TGT GAT CTG GGA GGC AAA-3' and reverse, 5'-CTC TTG CTG GAT GGG GAC A-3'; β-actin forward, 5'-CCT TCC TGG GCA TGG AGT CCT G-3' and reverse, 5'-GGA GCA ATG ATC TTG ATC TTC-3'. The PCR products were analyzed by melting curve analysis and agarose gel electrophoresis to determine product size and to confirm that no by-products were formed. The relative concentrations of the PCR products derived from the target gene were calculated using Applied Biosystems System software. The results were expressed relative to the number of β-actin transcripts used as an internal control. All experiments were conducted in triplicate.
Statistical analysis
The SPSS statistical software package version 16.0 (SPSS Inc., Chicago, IL, USA) was used for the analysis. All results were summarized as . Differences between groups were measured by two-tailed unpaired Student’s t-test. The correlation analysis between two sets of data was determined by Spearman’s correlation test. P<0.05 was considered statistically significant.
Results
Elevated Th22, Th17 cells and IL-22 in HCC
The frequencies of circulating Th22, Th17 cells were analyzed based on cytokine patterns after in vitro activation by PMA/ionomycin. The expression of a typical dot-plot of Th22, Th17 cells in HCC patients and healthy controls was shown in Figure 1A. The frequencies of Th22, Th17 cells were significantly elevated in HCC patients compared with those in healthy controls (Th22: 1.65%±0.14% vs. 0.39%±0.11%, P<0.001; Th17: 4.28%±1.55% vs. 2.19%±0.79%, P<0.001) (Figure 1B,C).
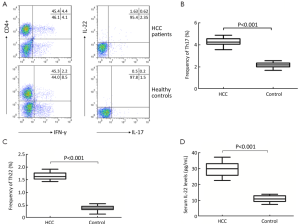
Similar to the observations in Th22 cells, the serum IL-22 levels in HCC patients were significantly elevated than those in healthy controls (29.85±3.98 vs. 11.09±1.83 pg/mL, P<0.001) (Figure 1D). Correlation analysis showed that the frequencies of Th22 cells were positively correlated to the serum IL-22 levels in HCC patients (r=0.338, P=0.023), but this correlation was not observed in healthy controls (r=0.029, P=0.907).
Th22 cell frequencies/serum IL-22 levels with hepatitis status
Because all the HCC patients in our study were hepatitis B virus (HBV) positive, we compared the Th22 cell frequencies and serum IL-22 levels according to the HBeAg status. However, we failed to show the difference in patients with HBeAg positivity or negativity in term of Th22 cell frequencies (1.74%±0.22% vs. 1.57%±0.46%, P=0.117) and serum IL-22 levels (30.54%±6.88% vs. 28.71%±5.52%, P=0.416).
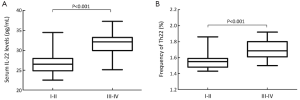
Th22 cells and serum IL-22 levels with TNM stage
The serum IL-22 levels at stage III-IV were higher than those at stage I-II (31.60%±3.21% vs. 27.44%±3.70%, P<0.001) (Figure 2A); and the similar results were observed with regard to the frequencies of Th22 cells, which were significantly elevated in HCC at stage III-IV compared with that at stage I-II (1.70%±0.13% vs.1.59%±0.14%, P=0.007) (Figure 2B).
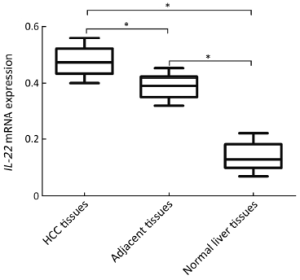
IL-22 protein and mRNA are elevated in HCC tissues
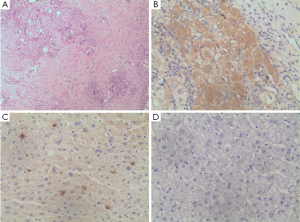
Immunohistochemical staining showed that IL-22 was located in non-parenchymal liver and IL-22-positive cells were distributed around the cancer nests. The distribution of IL-22 positive cells was highest in HCC tissues, followed by in adjacent HCC tissues and normal liver tissues (Figure S1).
The real-time PCR result showed that IL-22 mRNA expression was highest in HCC tissues, but lowest in normal liver tissues, and the expression in adjacent HCC tissues was between them (Figure 3). Moreover, higher IL-22 mRNA expression was found at stage III-IV than stage I-II of HCC tissues (0.46±0.05 vs. 0.19±0.05, P=0.028).
Discussion
T helper cells (Th) are a subgroup of lymphocytes, which play an important role in the immune system, particularly in the adaptive immune system. The Th subsets are defined according to their production of lineage-indicating cytokines and functions (12). Many Th cells, such as Th1, Th2 and Th17, function not only on other immune cells, but also on target tissue cells (13-15). Th22 cells are a new subset of Th cells, and distinct from other known T cell subsets. The phenotype of Th22 cells is CCR6+CCR4+CCR10+ and the key transcription factor is aryl hydrocarbon receptor (AHR). Th22 cells are similar to Th17 cells in their expression of chemokine (C-C motif) receptor 6 (CCR6) and CCR4. In contrast to Th17 cells, human Th22 cells express CCR10 but not CD161 (5). They express low or undetectable Th17- and Th1-associated transcription factors RORγt and T-bet (6). Th22 cells mediate their biological function via several cytokines, including IL-22, IL-26, and IL-13. Of which, IL-22 is the mainly functional cytokine, and the presence of AHR is required for its production. Recent studies indicate that IL-6 and TNF-α, along with the help of cytoid dendritic cells, can promote Th22 phenotype (5).
Th22 cells have been implicated in the pathogenesis of several autoimmune diseases, such as psoriasis and rheumatoid arthritis (16,17). In those studies, circulating Th22 cells were shown to elevate in the inflamed skin lesions and were positively correlated with disease severity. However, up to date, little data are available with regard to the role of Th22 cells in human cancers. Recent studies on the gastric cancer showed that the frequencies of circulating Th22 cells were elevated in patients compared with those of healthy controls, and also associated with the progression of gastric cancer (10,18). The elevated circulating Th22 cells were also reported in malignant pleural effusion compared with those of corresponding blood (11,19), suggesting that Th22 cells may be involved in the development of tumors. In our study, we compared the frequencies of Th22 cells in the peripheral blood of HCC patients and healthy controls, and found that the frequencies of Th22 cells were significantly elevated in HCC patients compared with those of healthy donors, suggesting that Th22 cells may be involved in T-cell-mediated immunity in HCC. In addition, we also found that frequencies of Th17 cells were higher in HCC patients than those of healthy donors, suggesting that Th22 cells may function together with Th17 cells in the pathogenesis of HCC. However, the synergistic effect of Th22 and Th17 cells in HCC remained unclear and needs further study.
IL-22 can be produced by several immune cells, including Th1, Th17, Th22 cells and NK cells. Among them, the production by Th22 cells accounts for 37% to 63% of total IL-22 (8). A previous study showed that the binding of IL-22 to it surface receptor in the rat hepatoma cell line H4IIE induced the rapid activation of JAK1 and Tyk2, leading to the phosphorylation of STAT-1, STAT-3 and STAT-5 (20). Production of IL-22 by immune cells reflects the activated role of immune cells, such as Th22, Th1, and Th17, on various chronic inflammatory diseases (21). The biological effects of IL-22 include increase of innate immunity, protection from damage as well as enhancement of regeneration. The target cells of IL-22 are certain tissue cells, including skin, liver and kidney, and from the respiratory organs to the gastrointestinal systems. The role of IL-22 can be either protective or pathogenic in chronic inflammatory diseases, which depends on the nature of the affected tissue and the local cytokine milieu (22).
The elevated serum IL-22 levels may participate in some T cell-mediated diseases. Some studies indicate that IL-22 is implicated in the pathogenesis of many autoimmune inflammatory diseases, including Crohn’s disease (9), psoriasis (16) and rheumatoid arthritis (17). Recently, Jiang et al. revealed that excessive IL-22 in the HCC microenvironment leads to growth of tumor, inhibition of apoptosis, and promotion of metastasis via STAT3 activation (23). In our study, we observed that serum IL-22 levels were significantly higher in HCC patients than in healthy controls. More importantly, we also observed that serum IL-22 levels were correlated with Th22 cells in HCC patients, but not in healthy controls. These observations are similar to the report of Jiang et al., and compatible with the idea that Th22 cells contribute to the pathogenesis of HCC through production of IL-22. Further, we observed that Th22 cells were increased as advance of tumor stage, suggesting that circulating Th22 cells may correlate with tumor growth and degree of HCC, but this correlation needs to be further studied by examining the expression of pSTAT3 or RORyt in HCC. In our study, we did not observed differences in patients with HBeAg positivity or negativity in term of Th22 cells frequencies and serum IL-22 levels, suggesting that Th22 cells/serum IL-22 may not be affect by the hepatitis status, however, due to the limitation of case number, this result should be confirmed in large sample studies.
It is well known that, in addition to the tumor-mediated immune modulation or immune-mediated tumor progression in cancer patients (24), many malignancies arise at the sites of inflammatory immune responses, and tumor-infiltrating lymphocytes as well as related cytokines they produced are accumulated in tumors region (25). IL-22 may have opposing short-term and long-term effects in the liver. Although IL-22 is able to protect mouse liver injury induced by concanavalin A and carbon tetrachloride (26), long-term expression of IL-22 during chronic inflammation may allow survival of damaged hepatocytes that are precursors of HCC, and therefore lead to tumorigenesis (27). In the present study, we next investigated the effects of IL-22 on the tumor microenvironment by detecting its protein and mRNA expression. The immunohistochemical staining showed that IL-22 was located at the sites of nonparenchymal liver; we speculated that HCC may secret some chemotactic factors to recruit Th22 cells from peripheral blood to the tumor region, subsequently Th22 cells producing IL-22 to perform the biological functions. However, this speculation needs further investigation. Similar to the observation of Jiang et al. (23), our results showed that IL-22 protein distribution and expression of IL-22 mRNA were higher around tumor region compare with adjacent HCC tissues and normal liver tissues. Furthermore, we also found that IL-22 protein and mRNA expression was higher at stage III-IV than at stage I-II of HCC, which was in line with the results of serum IL-22, implying that IL-22 was associated with tumor growth and degree of malignancy.
In summary, our results demonstrated that frequencies of circulating Th22 cells and IL-22 are significantly elevated in HCC patients compare with those of healthy controls, and they were elevated in advanced HCC patients. Further understanding the development, regulation and function of Th22 cells in HCC may provide insights for devising novel treatments for HCC.
Acknowledgements
This study was partially supported by research funding from the National Natural Science Foundation (No. 81260083) and National Natural Science Foundation (No. 31360221).
Disclosure: The authors declare no conflict of interest.
References
- Sia D, Villanueva A. Signaling pathways in hepatocellular carcinoma. Oncology 2011;81 Suppl 1:18-23. [PubMed]
- Wang H, Chen L. Tumor microenviroment and hepatocellular carcinoma metastasis. J Gastroenterol Hepatol 2013;28 Suppl 1:43-8. [PubMed]
- Harrington LE, Hatton RD, Mangan PR, et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol 2005;6:1123-32. [PubMed]
- Park H, Li Z, Yang XO, et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol 2005;6:1133-41. [PubMed]
- Duhen T, Geiger R, Jarrossay D, et al. Production of interleukin 22 but not interleukin 17 by a subset of human skin-homing memory T cells. Nat Immunol 2009;10:857-63. [PubMed]
- Eyerich S, Eyerich K, Pennino D, et al. Th22 cells represent a distinct human T cell subset involved in epidermal immunity and remodeling. J Clin Invest 2009;119:3573-85. [PubMed]
- Trifari S, Kaplan CD, Tran EH, et al. Identification of a human helper T cell population that has abundant production of interleukin 22 and is distinct from T(H)-17, T(H)1 and T(H)2 cells. Nat Immunol 2009;10:864-71. [PubMed]
- Wolk K, Witte E, Witte K, et al. Biology of interleukin-22. Semin Immunopathol 2010;32:17-31. [PubMed]
- Brand S, Beigel F, Olszak T, et al. IL-22 is increased in active Crohn’s disease and promotes proinflammatory gene expression and intestinal epithelial cell migration. Am J Physiol Gastrointest Liver Physiol 2006;290:G827-38. [PubMed]
- Liu T, Peng L, Yu P, et al. Increased circulating Th22 and Th17 cells are associated with tumor progression and patient survival in human gastric cancer. J Clin Immunol 2012;32:1332-9. [PubMed]
- Ye ZJ, Zhou Q, Yin W, et al. Interleukin 22-producing CD4+ T cells in malignant pleural effusion. Cancer Lett 2012;326:23-32. [PubMed]
- Zhang N, Pan HF, Ye DQ. Th22 in inflammatory and autoimmune disease: prospects for therapeutic intervention. Mol Cell Biochem 2011;353:41-6. [PubMed]
- Aujla SJ, Chan YR, Zheng M, et al. IL-22 mediates mucosal host defense against Gram-negative bacterial pneumonia. Nat Med 2008;14:275-81. [PubMed]
- Burgler S, Ouaked N, Bassin C, et al. Differentiation and functional analysis of human T(H)17 cells. J Allergy Clin Immunol 2009;123:588-95, 595.e1-7.
- Eyerich K, Pennino D, Scarponi C, et al. IL-17 in atopic eczema: linking allergen-specific adaptive and microbial-triggered innate immune response. J Allergy Clin Immunol 2009;123:59-66.e4.
- Kagami S, Rizzo HL, Lee JJ, et al. Circulating Th17, Th22, and Th1 cells are increased in psoriasis. J Invest Dermatol 2010;130:1373-83. [PubMed]
- Zhang L, Li JM, Liu XG, et al. Elevated Th22 cells correlated with Th17 cells in patients with rheumatoid arthritis. J Clin Immunol 2011;31:606-14. [PubMed]
- Zhuang Y, Peng LS, Zhao YL, et al. Increased intratumoral IL-22-producing CD4(+) T cells and Th22 cells correlate with gastric cancer progression and predict poor patient survival. Cancer Immunol Immunother 2012;61:1965-75. [PubMed]
- Ye ZJ, Zhou Q, Yuan ML, et al. Differentiation and recruitment of IL-22-producing helper T cells stimulated by pleural mesothelial cells in tuberculous pleurisy. Am J Respir Crit Care Med 2012;185:660-9. [PubMed]
- Lejeune D, Dumoutier L, Constantinescu S, et al. Interleukin-22 (IL-22) activates the JAK/STAT, ERK, JNK, and p38 MAP kinase pathways in a rat hepatoma cell line. Pathways that are shared with and distinct from IL-10. J Biol Chem 2002;277:33676-82. [PubMed]
- Witte E, Witte K, Warszawska K, et al. Interleukin-22: a cytokine produced by T, NK and NKT cell subsets, with importance in the innate immune defense and tissue protection. Cytokine Growth Factor Rev 2010;21:365-79. [PubMed]
- Rutz S, Ouyang W. Regulation of interleukin-10 and interleukin-22 expression in T helper cells. Curr Opin Immunol 2011;23:605-12. [PubMed]
- Jiang R, Tan Z, Deng L, et al. Interleukin-22 promotes human hepatocellular carcinoma by activation of STAT3. Hepatology 2011;54:900-9. [PubMed]
- Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 2011;331:1565-70. [PubMed]
- Albini A, Sporn MB. The tumour microenvironment as a target for chemoprevention. Nat Rev Cancer 2007;7:139-47. [PubMed]
- Pan H, Hong F, Radaeva S, et al. Hydrodynamic gene delivery of interleukin-22 protects the mouse liver from concanavalin A-, carbon tetrachloride-, and Fas ligand-induced injury via activation of STAT3. Cell Mol Immunol 2004;1:43-9. [PubMed]
- Park O, Wang H, Weng H, et al. In vivo consequences of liver-specific interleukin-22 expression in mice: Implications for human liver disease progression. Hepatology 2011;54:252-61. [PubMed]